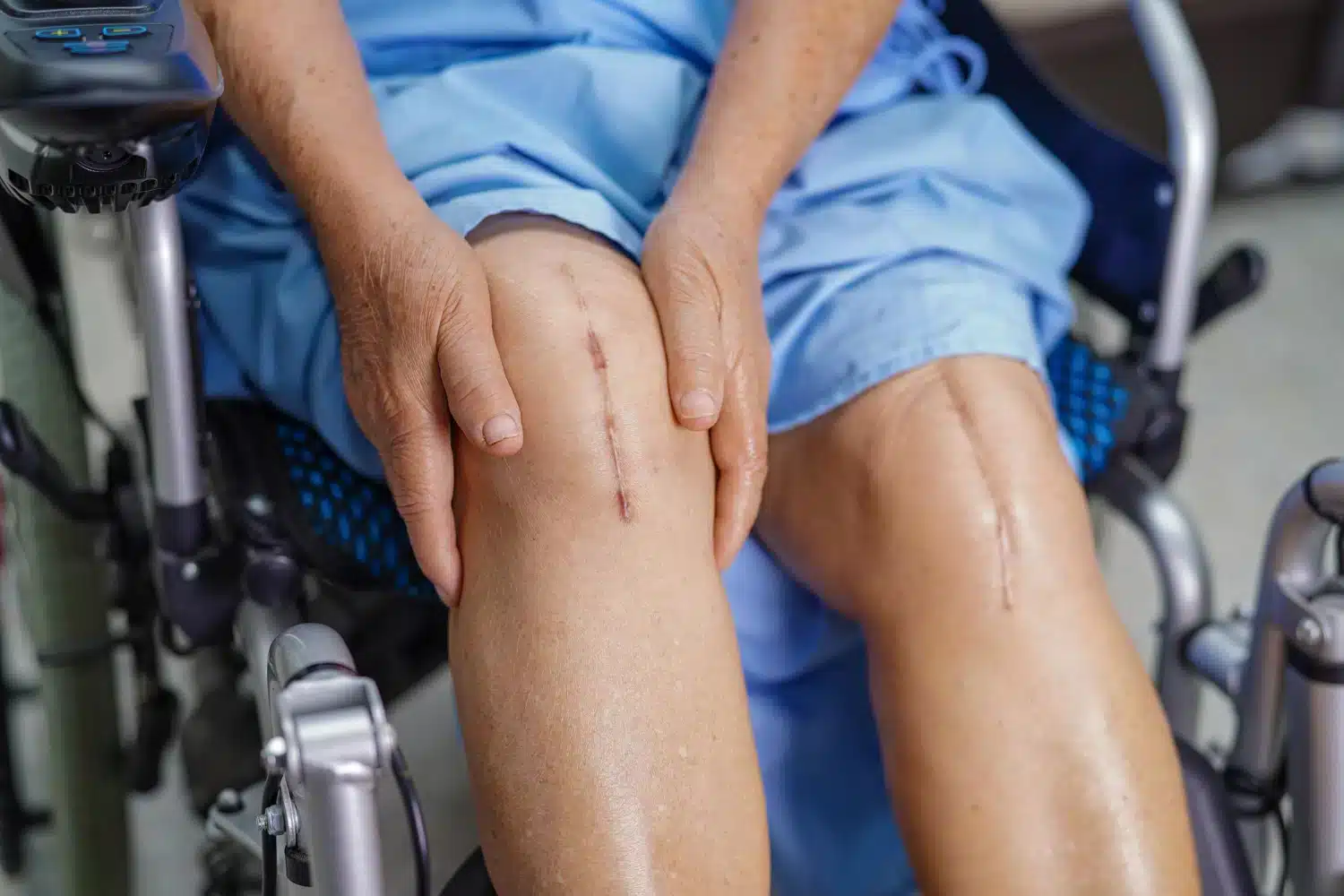
Defective Medical Devices
Aggressive Texas Defective Drugs & Devices Attorney
24/7 FREE Consultation with a Defective Drugs & Devices Lawyer. We’ve won MILLIONS for our clients over 20 years. No Fees Unless We Win.
Free Consultation 24/7 Nosotros Hablamos Español
Unveiling the Dangers: The Reality Behind Defective Medical Devices
In the world of modern medicine, the use of technology and devices has revolutionized patient care. However, when these devices are flawed or malfunction, they can pose serious risks to patients. We will delve into the disturbing truth behind the problems associated with defective medical devices.
From hip replacements to pacemakers, medical devices are designed to improve and prolong lives. But what happens when these devices fail? The consequences can be devastating. Patients may experience severe pain, complications, and even life-threatening situations. It is crucial to shed light on this issue to protect patients and encourage accountability within the medical device industry.
We will examine real-life case studies, government regulations, and industry practices related to defective medical devices. We will explore the reasons behind these defects, the impact on patients and their families, and what can be done to prevent future tragedies.
Join us as we lift the veil on the hidden dangers of defective medical devices and shine a light on the reality that patients and healthcare providers must confront.
Common types of defective medical devices
Medical devices are designed to improve and enhance patient care, but when they are defective, they can pose serious risks to the health and well-being of individuals. Some of the most common types of defective medical devices include hip and knee replacements, pacemakers, defibrillators, surgical mesh, and breast implants.
Hip and knee replacements are designed to provide relief from chronic pain and improve mobility, but when they are defective, they can cause severe pain, swelling, and even the need for revision surgery. Pacemakers and defibrillators are essential for managing heart conditions, but if they malfunction, they can fail to regulate the heart’s rhythm, leading to life-threatening complications.
Surgical mesh, used to repair hernias and pelvic organ prolapse, has been linked to a range of complications, including infection, organ perforation, and chronic pain. Breast implants, while primarily used for cosmetic purposes, can also pose health risks if they rupture or leak, leading to inflammation, scarring, and other complications.
These are just a few examples of the types of defective medical devices that have been the subject of widespread attention and legal action. As technology continues to advance, it is essential that medical device manufacturers and regulatory bodies ensure the safety and efficacy of these products to protect patients from harm.

The impact of defective medical devices on patients
The impact of defective medical devices on patients can be devastating, both physically and emotionally. Patients who have been harmed by these devices often experience a range of complications, including chronic pain, disability, and even life-threatening conditions.
In many cases, the consequences of a defective medical device can be long-lasting and far-reaching. Patients may require multiple surgeries, extended hospital stays, and ongoing medical treatment to address the issues caused by the device. This can lead to significant financial and personal burdens, as well as emotional distress and a reduced quality of life.
Beyond the physical and financial toll, defective medical devices can also have a profound impact on a patient’s mental and emotional well-being. The trauma of experiencing a medical complication, the uncertainty of their future health, and the frustration of navigating the legal and healthcare systems can all take a heavy toll on a patient’s mental health. Many patients report feelings of anxiety, depression, and even post-traumatic stress disorder as a result of their experiences.
It is important to recognize the far-reaching consequences of defective medical devices and the impact they can have on patients and their families. By understanding the full scope of the problem, we can work towards more effective solutions and greater accountability within the medical device industry.
The role of regulatory bodies in monitoring medical devices
Regulatory bodies play a crucial role in monitoring the safety and efficacy of medical devices. In the United States, the Food and Drug Administration (FDA) is the primary agency responsible for overseeing the medical device industry, ensuring that products meet strict safety and performance standards before they can be marketed and sold.
The FDA’s regulatory process involves a thorough review of medical device applications, including clinical trials and post-market surveillance data. The agency also has the authority to issue recalls and safety alerts when defective or potentially dangerous devices are identified. However, the effectiveness of the FDA’s oversight has been the subject of ongoing debate, with critics arguing that the agency’s processes are not stringent enough to prevent the widespread distribution of defective devices.
In addition to the FDA, other regulatory bodies, such as the Centers for Medicare and Medicaid Services (CMS) and the Consumer Product Safety Commission (CPSC), also play a role in monitoring medical devices. These agencies may investigate reports of device-related injuries or deaths and take appropriate action to protect public health and safety.
Despite these regulatory efforts, the reality is that defective medical devices continue to pose a significant risk to patients. This highlights the need for stronger oversight, more rigorous testing and approval processes, and greater transparency and accountability within the medical device industry. Patients and healthcare providers must remain vigilant and advocate for more effective regulatory measures to ensure the safety and well-being of all those who rely on these critical medical technologies.

Legal implications of defective medical devices
When patients are harmed by defective medical devices, they may have legal recourse to seek compensation and hold the responsible parties accountable. Defective medical device lawsuits can be complex and challenging, but they play a crucial role in promoting product safety and deterring manufacturers from prioritizing profits over patient safety.
The legal theories underlying defective medical device cases can vary, but they often include claims of design defects, manufacturing defects, and failure to warn. Patients may be able to seek damages for their medical expenses, lost wages, pain and suffering, and other related costs. In some cases, class-action lawsuits may be filed on behalf of a group of affected individuals.
The success of these legal actions can depend on a variety of factors, including the strength of the evidence, the expertise of the legal team, and the specific laws and regulations governing medical devices in the jurisdiction. Navigating the legal system can be daunting for patients and their families, but it is essential to hold manufacturers and healthcare providers accountable for the harm caused by defective devices.
In addition to individual lawsuits, regulatory bodies and government agencies may also take legal action against medical device manufacturers. This can include fines, product recalls, and even criminal charges in cases of egregious misconduct or negligence. These legal interventions play a critical role in deterring future violations and promoting a culture of safety and accountability within the medical device industry.
Steps to take if you have been harmed by a defective medical device
If you or a loved one have been harmed by a defective medical device, it is important to take immediate action to protect your rights and seek the appropriate medical and legal assistance. Here are some steps you can take:
- Seek medical attention: Your first priority should be to address any immediate health concerns and receive the necessary medical care. This may involve additional procedures, treatments, or surgeries to address the issues caused by the defective device.
- Document the issue: Keep detailed records of your medical treatment, including any complications or side effects you have experienced. This documentation can be crucial evidence in a potential legal case.
- Report the issue: Notify the manufacturer of the defective device and the FDA about the problem. This helps to ensure that the issue is properly documented and that appropriate action can be taken to address the safety concerns.
- Consult with a medical device attorney: Consider speaking with a lawyer who specializes in defective medical device cases. They can provide guidance on your legal options and help you navigate the complex process of pursuing a claim against the manufacturer.
- Participate in any recalls or safety alerts: If the defective device has been the subject of a recall or safety alert, it is important to follow the instructions provided and participate in any necessary corrective actions.
- Seek support: The physical, emotional, and financial toll of dealing with a defective medical device can be overwhelming. Seek support from family, friends, and patient advocacy groups to help you cope with the challenges you may be facing.
By taking these steps, you can protect your rights, seek the compensation you deserve, and contribute to the ongoing efforts to improve the safety and accountability of the medical device industry.

Case studies of high-profile defective medical device lawsuits
The legal landscape surrounding defective medical devices is filled with high-profile cases that have had significant impacts on patients, the industry, and the legal system. Here are a few examples:
- DePuy ASR Hip Replacement Recall: In 2010, Johnson & Johnson’s DePuy Orthopaedics division recalled its ASR hip replacement system due to a high failure rate. Thousands of patients suffered complications, including pain, swelling, and the need for revision surgery. This led to a series of lawsuits, resulting in over $2.5 billion in settlements for affected patients.
- Transvaginal Mesh Litigation: Transvaginal mesh devices, used to treat pelvic organ prolapse and stress urinary incontinence, have been the subject of numerous lawsuits due to a range of complications, including pain, organ perforation, and the need for additional surgeries. Manufacturers, such as Johnson & Johnson and Boston Scientific, have faced billions of dollars in settlements and court judgments.
- Medtronic Infuse Bone Graft Lawsuits: The Medtronic Infuse Bone Graft, a device used in spinal surgeries, has been linked to a range of complications, including cancer, sterility, and nerve damage. Hundreds of lawsuits have been filed against Medtronic, alleging that the company failed to disclose the risks associated with the device.
These cases illustrate the significant impact that defective medical devices can have on patients and the importance of holding manufacturers accountable. They also highlight the need for more stringent regulations, improved post-market surveillance, and greater transparency within the medical device industry to protect patient safety.
The importance of product recalls and safety alerts
Product recalls and safety alerts play a crucial role in addressing the issue of defective medical devices and protecting patients from harm. When a medical device is found to be defective or potentially dangerous, the manufacturer or regulatory bodies, such as the FDA, may issue a recall or safety alert to inform healthcare providers and patients about the issue and the necessary steps to be taken.
Recalls can range from the removal of a specific lot or batch of a device to the complete withdrawal of a product from the market. Safety alerts, on the other hand, may provide information about potential risks or complications associated with a device, even if a full recall is not warranted. Both of these mechanisms are designed to prevent further harm and encourage the prompt identification and resolution of issues.
However, the effectiveness of these measures is often limited by the challenges of reaching all affected patients and ensuring that appropriate actions are taken. Patients may not be aware of a recall or safety alert, or they may face barriers in accessing the necessary information or resources to respond to the issue. Additionally, some manufacturers may be reluctant to issue recalls or provide transparent information about device-related problems, prioritizing their financial interests over patient safety.
To address these challenges, it is essential to improve the communication and dissemination of recall and safety alert information, as well as to strengthen the regulatory frameworks that govern the medical device industry. By ensuring that patients and healthcare providers are informed and empowered to respond to defective devices, we can enhance the overall safety and effectiveness of medical technologies and protect the well-being of those who rely on them.
How to stay informed and protect yourself from defective medical devices
In the face of the ongoing challenges posed by defective medical devices, it is crucial for patients and healthcare providers to stay informed and take proactive steps to protect themselves. Here are some strategies to help you stay informed and safeguard your health:
- Research medical devices: Before undergoing a procedure that involves a medical device, take the time to research the device, its manufacturer, and any known issues or recalls. This information can help you make informed decisions about your healthcare and advocate for your own safety.
- Stay up-to-date on recalls and safety alerts: Regularly check the FDA’s website and other reputable sources for information on medical device recalls and safety alerts. Sign up for email or text notifications to receive timely updates on any issues that may affect you or your loved ones.
- Communicate with your healthcare provider: Discuss your concerns about medical devices with your healthcare provider, and work together to weigh the risks and benefits of any proposed treatments or procedures. Your provider should be willing to address your questions and provide you with the information you need to make an informed decision.
- Join patient advocacy groups: Consider joining patient advocacy groups or organizations that focus on medical device safety. These groups can provide valuable resources, support, and opportunities to get involved in efforts to improve device safety and accountability.
- Report issues: If you experience any complications or side effects related to a medical device, report the issue to the manufacturer and the FDA. Your report can help to identify and address emerging problems, potentially preventing harm to other patients.
By staying informed, communicating with healthcare providers, and taking an active role in your own healthcare, you can help to protect yourself and contribute to the ongoing efforts to improve the safety and reliability of medical devices.
Why McKay Law?

Passion For Justice – Compassion For Clients
Compassion. Confidence. Commitment.
20+ years of combined experience with of counsel addition.
Reasons to Hire McKay Law
VIP / White Glove Service
We are Available 24/7 - We take care of our client’s needs – whatever they may be – and advise and instruct from beginning to end.
We Care
We hope to change your life and your situation and turn something tragic into something better. No issue is too tiny for us to consider or too big for us to tackle.
We Listen to Learn Your Story
Injuries, pain, the stress of life, the case, finances – We want to know you and be the voice to tell your story for the best results.
Any Legal Issue
We are Texas Tough to help you. If we can’t address, we know the best.
Fight For Best Results
We ensure we fight for the best results possible for your case.
No Fee
Unless we win and also NO FEE for handling property damage.
20+ Years of Combined Experience
We have a long line of success and a team that has worked together for decades and has perfected and streamlined the process; making something stressful and painful as seamless and easy as possible.
Dedicated Nurse to Guide Medical Treatment
McKay Law has an in-house specialist guiding our personal injury clients on medical treatment. Read more about our Texas Nursing Board Certified Nurse and How We Assist.
Current Mass Tort Lawsuits
Asbestos & Mesothelioma
McKay Law Client Reviews on Google
Free Consultation with Texas Mass Tort Lawyer
No Fees Unless We Win. We Listen To Our Clients.
Lindsey McKay is a dedicated trial attorney with experience and drives to get the best results for her clients. We handle Mass Tort cases that involve the following matters: Asbestos & Mesothelioma, Drugs & Devices, Environmental, and Consumer.
















Free Consultation 24/7 Nosotros Hablamos Español
You may be owed money.
Call our Personal Injury Lawyer now: (903) 465-8733 or use the form below. You can also calculate your total damages for settlements.